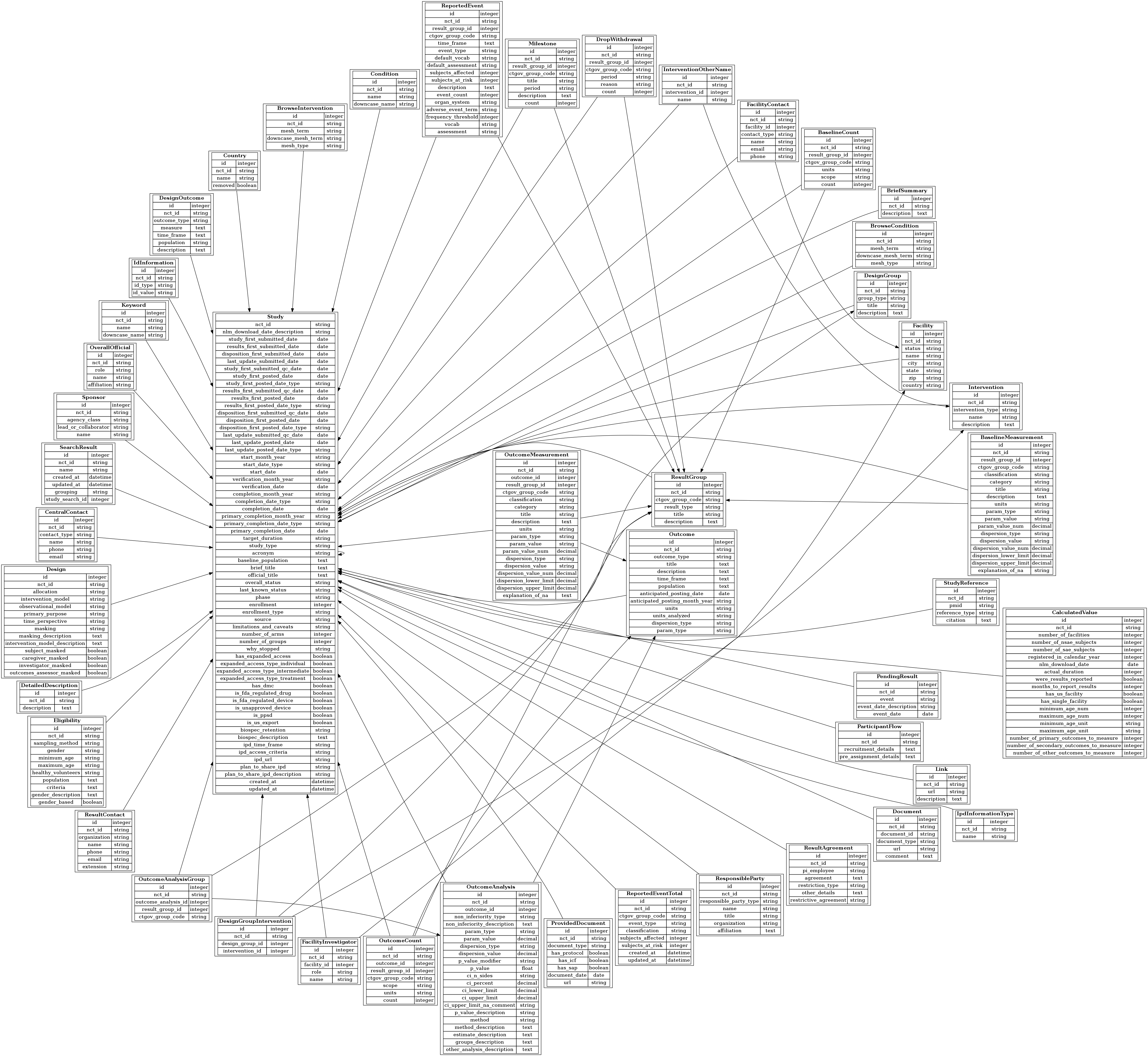
The Database for Aggregate Analysis of ClinicalTrials.gov (AACT) and Subsequent Regrouping by Clinical Specialty | PLOS ONE
Discrepancies between FDA documents and ClinicalTrials.gov for Orphan Drug-related clinical trial data | PLOS Global Public Health

Characteristics of clinical trial websites: information distribution between ClinicalTrials.gov and 13 primary registries in the WHO registry network – topic of research paper in Clinical medicine. Download scholarly article PDF and read

PDF) Update on the clinical trial landscape: analysis of ClinicalTrials.gov registration data, 2000–2020

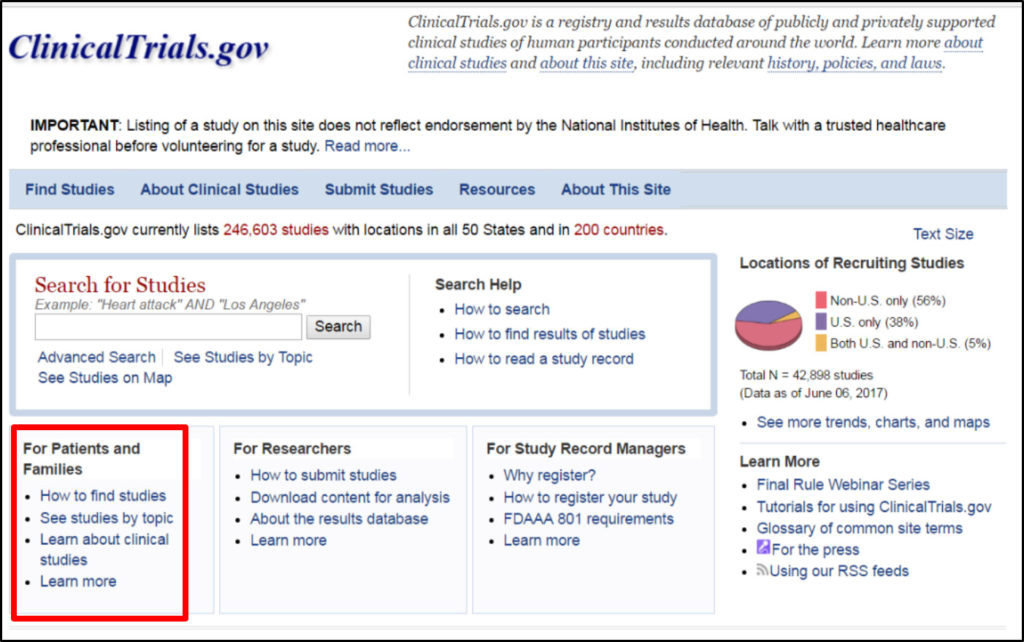
Alliance for Aging Research Suggests Improvements to ClinicalTrials.gov - Alliance for Aging Research
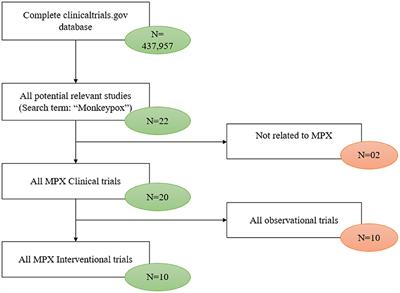
Frontiers | Characterization of interventional clinical trials for monkeypox; systematic review of ClinicalTrials.gov database